StrokeDX
StrokeDX
Revolutionizing stroke diagnostics
TL;DR
The solution is: Built the software and analytics platform powering a portable, non-invasive handheld stroke diagnostic device, enabling real-time cerebral blood flow visualization at the point of care
Engineered the full communication system for hardware data acquisition and control, including USB protocol integration to support a new generation of the STEDI device
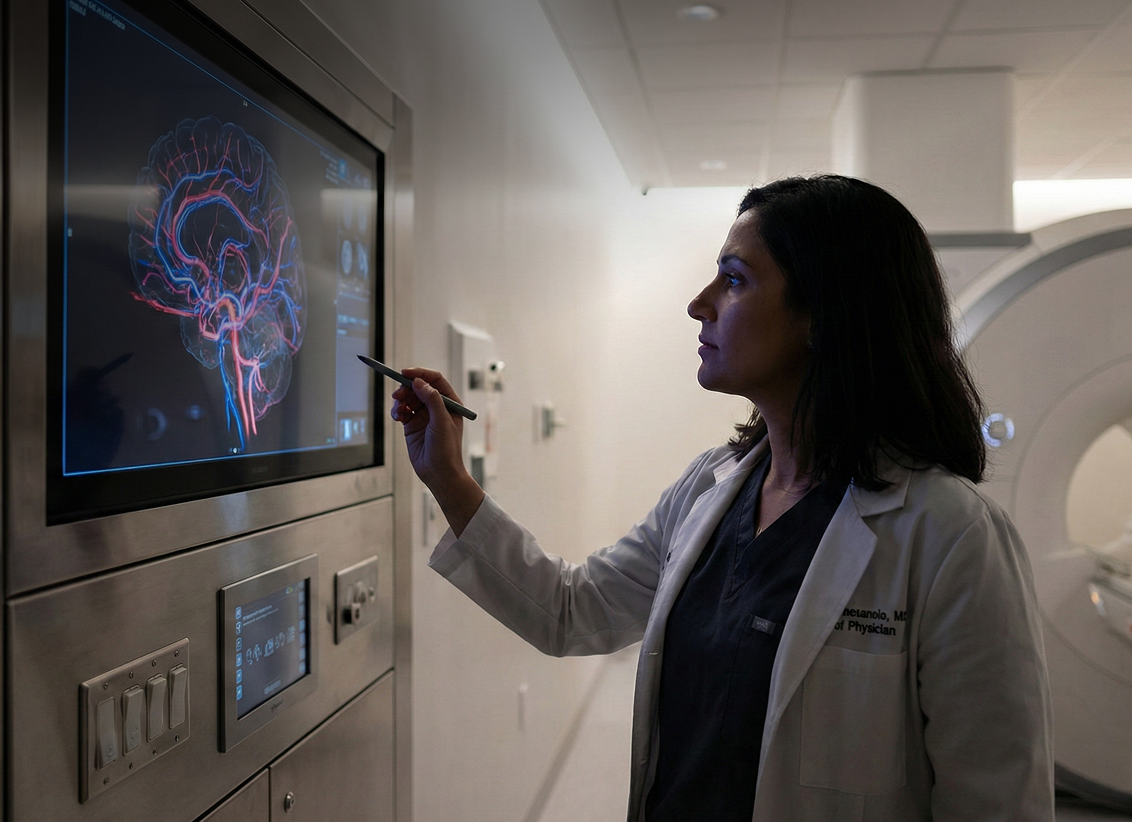
Designed a clinical-grade interface for emergency settings with real-time signal processing to detect and differentiate ischemic and hemorrhagic stroke signatures
The Challenge
Every minute of a stroke goes untreated, an estimated 1.9 million neurons die. Speed of diagnosis is not a clinical nicety; it is the primary determinant of whether a patient survives with their faculties intact. Yet the gold standard for stroke diagnosis, CT and MRI imaging, requires transporting the patient to a hospital, waiting for machine availability, and having a radiologist interpret results. In emergency settings outside major hospital centers, and especially in underserved regions, this chain of dependencies routinely costs patients the critical window for effective intervention.
StrokeDX was developing a fundamentally different approach: a portable, handheld, non-invasive cerebrovascular monitoring device that physicians and EMTs could use at the point of care. The hardware used non-radiating eddy current sensors to detect changes in cerebral blood flow, producing signal data that could differentiate ischemic strokes (blockages) from hemorrhagic strokes (bleeds), a critical distinction because the treatments for each are opposite.
The hardware innovation was only half the equation. Raw sensor data means nothing without software that can acquire it reliably, process it in real time, and present it in a form that a clinician under pressure can act on in seconds. StrokeDX needed a software partner who could build a clinical-grade platform, one capable of controlling the device, handling the data pipeline, running advanced signal analytics, and displaying results through an interface designed for high-stress emergency use.
The stakes for getting this right extended well beyond the engineering brief. Software failures in a diagnostic device are not bugs to be patched in the next release; they are potential patient safety events. Every architectural decision needed to account for clinical-grade data integrity, HIPAA compliance, and the eventual pathway to FDA regulatory clearance.
Key Results
Real-time cerebral blood flow visualization platform delivered for point-of-care emergency use
Signal analytics pipeline differentiating ischemic and hemorrhagic stroke signatures
Multi-generation hardware support: original device through USB-based next-gen STEDI integration
Clinical-grade data pipeline with HIPAA-compliant secure storage in SDX format
StrokeDX raised nearly $10M in funding; won MedTech Innovator Global Competition 2023
Frequently Asked Questions
Published: · Last updated:
Ready to build something amazing?
Let's discuss how we can help transform your ideas into reality.